- Home
- News
- Spotlight on Science
- Creating reactivity...
Creating reactivity using unstable endmembers
05-02-2016
A new synthetic approach was used to create a solid solution of Mg and Fe nitrides bringing together unreactive starting materials and using pressure to remove the barrier to reactivity. This approach opens up new synthetic possibilities across the periodic table involving combinations of materials that normally would not be considered.
A pivotal attribute to preparing sought-after new materials and material landscapes is reactivity. An accompanying principal challenge is to have control over which structure is formed. In a solid solution, the crystal structure is generally dictated by that of the endmembers. But there is a problem, however, for a solid solution where the starting materials do not react and even the endmembers that would dictate formation of the desired structure do not exist. Here, using pressure, we have removed barriers to reactivity by making the source of solid solution end-member instability the driving force for solid solution stability in the desired structure.
Many materials are solid solutions, chemically homogeneous combinations of two (or more) constituents, where the atoms or ions of one replace those of the other within the same crystal structure. The members of a solid solution series will differ in the proportions of their constituents, with endmembers being the individual constituents (starting materials) at the extremities of a series. In general, for a solid solution to form, the endmembers should have the same native crystal structure and similar unit cell volume.
Our study focused on magnesium-iron nitride. A principal technological importance of Mg is that it is the lightest metal used in structural materials. Thus, combining Mg with heavier metals has significant structural, energetic and biomechanical advantages [1]. A particular incentive for this nitride synthesis is due to the intriguing potential application of MgN and FeN systems in spintronics, where electron spin is exploited for information processing and storage [2].
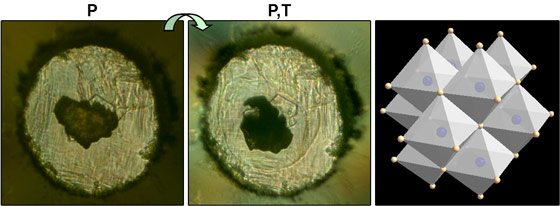
However, for magnesium-iron nitride, there is a significant difference in size (lattice parameter) between the endmembers MgN and FeN preventing reactivity. Moreover, these two endmembers do not exist in the desired bulk crystal structure (rocksalt, Fm3m) at ambient or elevated pressures. Our approach was to use high pressure, between 15 and 23 GPa, along with nitrogen incorporation. The high pressure exerts a greater effect on the more compressible Mg so removing the endmember lattice mismatch. Next, high temperature, 2500 K, permits a stabilising redistribution of valence electrons between FeN and MgN. Using this approach, we have made a new rocksalt Mg0.4Fe0.6N solid solution (Figure 1). This is a system where neither a Mg-Fe alloy nor a Mg-Fe-N nitride solid solution can be made at ambient pressure.
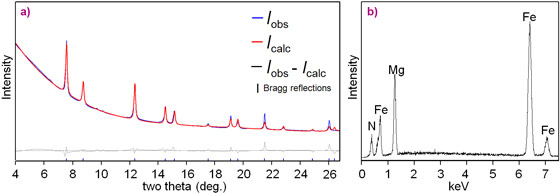
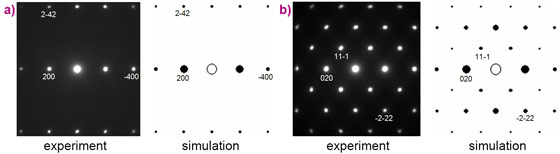
For the chemical synthesis we used both laser-heated diamond anvil cells and multi-anvil methods. The samples for analysis were small. Characterisation was facilitated by using beamline ID11 where we could measure synchrotron X-ray diffraction (Figure 2) together with fluorescence from the same place in the sample and link the crystal structure with the chemical composition. This data was combined with precession electron diffraction (Figure 3), scanning and field emission scanning electron microscopy with energy and wavelength dispersive analysis. All together, these techniques gave us a high resolution structural, chemical and morphological characterisation of the reaction products [3].
In addition to its technological significance, rocksalt MgxFe1-xN has crystal chemical and planetary science importance because it is isostructural to MgxFe1-xO, which comprises 20% by volume of the lower mantle, and because of the nitrogen concentration in the deep Earth, recently reported to be greater than previously thought [4]. Hence, the synthesis and characterisation of this sample lays a foundation for addressing the effects that light elements (e.g. N, C) can have on alloying of light metals, including Mg, with Fe on the composition and density of the Earth’s core.
Finally, we suggest that the materials chemistry result here, where the desired bulk rocksalt structure is not achievable by the two end-members in isolation, but only in the context of each other, has some analogy to mathematical developments in game theory, where an optimal result is reached only when players do what is best for them and for the other member [5]. This latter type of formalism may also become a tool in predicting materials behaviour and outcome.
Principal publication and authors
Creating reactivity with unstable endmembers using pressure and temperature: synthesis of bulk cubic Mg0.4Fe0.6N, G. Serghiou (a), G. Ji (b), N. Odling (c), H.J. Reichmann (d), J.P. Morniroli (e), R. Boehler (f), D.J. Frost (g), J.P. Wright (h), B. Wunder (d), Angew. Chem. Int. Ed. 54, 15109 (2015); doi: 10.1002/anie.201506257.
(a) School of Engineering, University of Edinburgh (UK)
(b) Unité Matériaux et Transformations, UMR CNRS 8207, Université Lille 1 (France)
(c) School of Geosciences, University of Edinburgh (UK)
(d) Helmholtz-Zentrum Potsdam, German Research Centre for Geosciences (Germany)
(e) Université Lille 1 and Ecole Nationale Supérieure de Chimie de Lille (France)
(f) Geophysical Laboratory, Carnegie Institution of Washington (USA)
(g) Bayerisches Geoinstitut, Universität Bayreuth (Germany)
(h) ESRF
References
[1] R.H. Taylor, S.H. Curtarolo, G.L.W. Hart, Phys. Rev. B 84, 084101 (2011).
[2] A. Droghetti, N. Baadji, S. Sanvito, Phys. Rev. B 80, 235310 (2009).
[3] G. Serghiou, G. Ji, M. Koch-Müller, N. Odling, H.J. Reichmann, J.P. Wright, P. Johnson, Inorg. Chem. 53, 5656 (2014).
[4] B. Marty, Earth Planet. Sci. Lett. 313, 56 (2012).
[5] J. F. Nash, Proc. Natl. Acad. Sci USA 36, 48 (1950).
Top image: Exploiting the instability of solid solution endmembers to create new materials under pressure.