- Home
- News
- Spotlight on Science
- Spatially quantifying...
Spatially quantifying crystallographic heterogeneities in operating Li-ion electrodes
21-02-2020
The performance of electrodes in Li-ion batteries is impeded by unfavourable chemical heterogeneities that pre-exist or develop during operation. Such heterogeneities were spatially and temporally quantified using high-resolution X-ray diffraction-computed tomography, revealing new insights into the onset of degradation and means to improve the performance of Li-ion cells.
Li-ion batteries are the technology of choice for energy storage applications that require a cost-effective and energy-dense solution, with the greatest demand coming from electrified transport. Improving the performance and safety of Li-ion batteries is imperative for the widespread uptake of electric vehicles. One promising approach is to more finely control the stoichiometry and morphology of the active materials within cells. Current electrode materials contain particles consisting of transition metal oxides, such as LixMn2O4 where 0 < x ≤ 1, and are known to be unfavourably heterogeneous in both chemistry and morphology. Dynamic chemical and structural heterogeneities across multiple length scales are known to lead to battery degradation and failure [1]. For example, strain-induced cracking of electrode particles can stem from lithiation gradients and cause impedance growth [2], transition metal dissolution can lead to irreversible capacity loss at both the positive and negative electrodes [3], and spatially-dependent rates of lithiation can lead to underutilisation of capacity [4]. Quantifying crystallographic heterogeneities in large representative volumes as they change during operation with resolutions sufficient for sub-particle measurements is highly desirable to achieve insight into inter- and intra-particle phenomena.
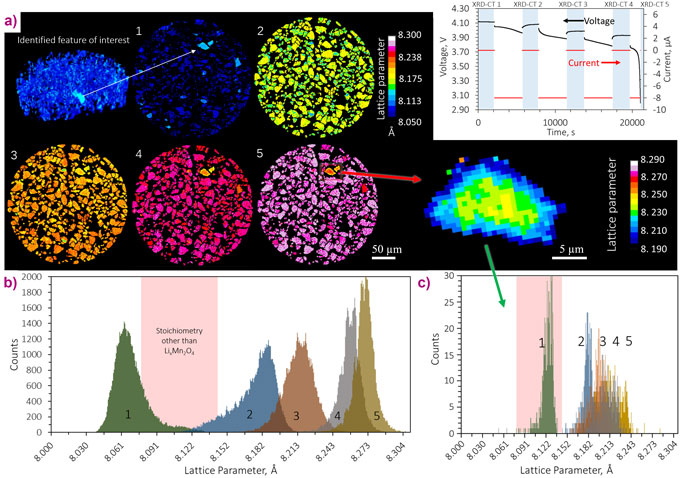
X-ray diffraction-computed tomography (XRD-CT) facilitates non-destructive 3D crystallographic quantification of chemical heterogeneities within a sample material. With recent advances in synchrotron brilliance, detector capabilities, and data processing strategies, high resolution XRD-CT is now possible with submicrometre resolution over short periods of time. At beamline ID15A, high-resolution (1 µm) XRD-CT scans of a LixMn2O4 electrode were taken periodically during operation to understand stoichiometric heterogeneities between particles and radial variations of stoichiometry and phase mass fractions within single particles (Figure 1).
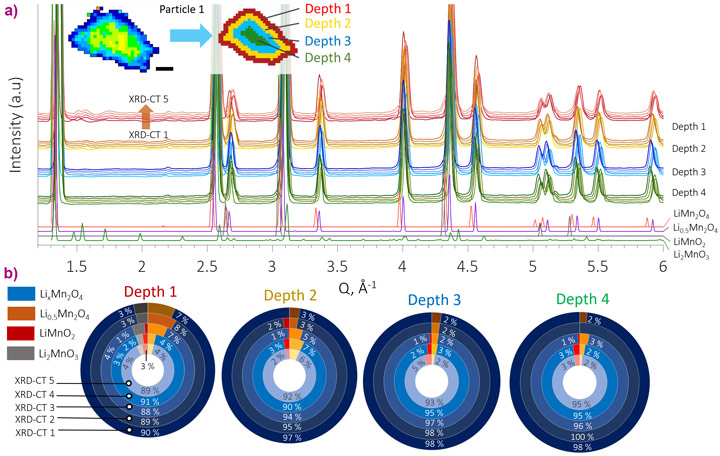
In Figure 1b, a histogram of the lattice parameter for all particles as they were lithiated shows that there was a wide distribution of lattice parameter within and between electrode particles. It was shown that the response of distinct particles to lithiation varied widely throughout the electrode. For example, the behaviour of the particle isolated in Figure 1a deviated from the expected behaviour of LixMn2O4. This particle was further investigated independently from the rest, and its histogram in Figure 1c showed that it contained a lattice parameter that was not typical of LixMn2O4. The particle was segmented into different depth regions in the XRD-CT reconstructions such that depth profiling of phase mass fractions could be carried out (Figure 2). Depth profiling revealed a stoichiometric gradient within the particle, with higher concentrations of Li2MnO3 near the surface. This phase is resistant against Mn dissolution into the electrolyte, thus likely protecting the particle from further degradation, but it is also lower in capacity than LixMn2O4, which would negatively affect the cell’s energy density. In the bulk electrode, the intermediate Li0.5Mn2O4 phase was identified mid-way through the lithiation process, and rock salt LiMnO2 was shown to form at every depth in most particles during lithiation, even at low states of lithiation during the first cycle of the cell. LiMnO2 has a higher diffusion coefficient than LixMn2O4 and its presence would negatively affect the rate performance of the cell. A separate electrode sample from the same batch of material was cycled 150 times and the sample was then imaged ex-situ. The concentration of phases associated with degradation also varied spatially between particles, indicating that not all particles degraded equally.
XRD-CT allowed amplification of signal from specific phases of interest by segmenting and distinctly quantifying phase fractions from regions where their presence was highest; using conventional point XRD measurements, such detail would likely be lost in the noise. The wide spatial distribution of phases present at the beginning of the electrode’s life and after extensive cycling demonstrates that there is opportunity for manufacturers to optimise synthesis techniques to produce a more uniform batch of electrode material with favourable stoichiometries. Fine control of particle stoichiometry would improve the lifetime, safety and performance of Li-ion cells. Future work on determining the efficacy of novel synthesis techniques to produce uniform electrodes, and electrodes with spatial stoichiometries that are resistant to known degradation mechanisms, can be validated with XRD-CT and will guide the design of next generation Li-ion battery materials.
Principal publication and authors
Spatial quantification of dynamic inter and intra particle crystallographic heterogeneities within lithium ion electrodes, D.P. Finegan (a), A. Vamvakeros (b,c,d), C. Tan (e,f), T.M.M. Heenan (d,e), S.R. Daemi (d,e), N. Seitzman (f), M. Di Michiel (b), S. Jacques (c), A.M. Beale (c,d,g), D.J.L. Brett (d,e), P.R. Shearing (d,e) & K. Smith (a), Nature Communications 11, 631 (2020); doi: 10.1038/s41467-020-14467-x.
(a) National Renewable Energy Laboratory, Golden (USA)
(b) ESRF
(c) Finden Limited, Abingdon (UK)
(d) University College London (UK)
(e) The Faraday Institution, Didcot (UK)
(f) Colorado School of Mines, Golden, (USA)
(g) Rutherford Appleton Laboratories, Didcot (UK)
References
[1] S.J. Harris, P. Lu, The Journal of Physical Chemistry C 117, 6481-6492 (2013).
[2] Y. Mao et al., Advanced Materials 29, 1900247 (2019).
[3] C. Zhan et al., Nature Communications 4, 2437 (2013).
[4] D.P. Finegan et al., Advanced Science 3, 1500332 (2016).