- Home
- News
- Spotlight on Science
- High-spatial-resolution...
High-spatial-resolution 3D imaging of human spinal cord and column anatomy
08-12-2020
Modern high-spatial-resolution radiologic methods are enabling increasingly detailed volumetric postmortem investigations of human neuroanatomy for diagnostic, research, and educational purposes. X-ray phase-contrast micro-CT on beamline ID17 uniquely renders both hard-tissue vertebral bone structure and soft-tissue spinal cord neuroanatomy in 3D, including deep microscale cellular and vascular features, without contrast agent use.
Detailed visualisation of human neuroanatomy is paramount to a better understanding of the structure, function and diseases of the human central nervous system, and advancements in both in vivo and postmortem neuroimaging have had a wide impact on modern medicine, from diagnostics and public health management to the assessment of disease etiology and educational purposes [1]. Emerging technologies for dissection-free postmortem microscale 3D analysis of fixed soft-tissue human central nervous system specimens include high-field-strength postmortem MRI [2] and tabletop absorption-based micro- to nano-CT systems [3].
Complementary to MRI microscopy, and more sensitive and efficient than absorption-based micro-CT, synchrotron-based X-ray phase-contrast micro-CT is a tissue-conserving, contrast-agent-free technique, which provides wide fields of view, time-efficient measurements and high soft-tissue sensitivity for microscale 3D imaging of large fixed human organ specimens, including human breast samples [4], human carotid artery specimens [5] and human knee joints [6]. At the price of reduced fields of view, cutting-edge synchrotron setups can also reach submicron spatial resolutions, and this methodology can be applied to diverse animal model neuroimaging work [7-9]. Here, the 3D anatomy of the human spine, a challenging organ to image owing to the thick and complex bone structures surrounding the spinal cord environment, was studied at high spatial resolution using synchrotron-based phase-contrast micro-CT at beamline ID17.
Several multi-vertebral human spinal column samples, unilaterally perfused with an iodinated vascular contrast agent, were harvested from Thiel-embalmed human cadavers. Thiel embalming involves cadaver perfusion and immersion for several months in a fluid made up mainly of water, glycol, and various salts, used by medical schools for educational purposes. Moreover, other human spinal cord samples were extracted from the bony intervertebral canal and formalin-fixed. All samples were imaged with monochromatic 60-keV X-rays and a propagation-based X-ray phase-contrast micro-CT with 463 μm3 and 83 μm3 voxel sizes. Phase-retrieved CT datasets were reconstructed and rendered in 3D after segmentation of hard versus soft tissues.
Fig. 1: a) Sagittal, (b) axial and (c) zoomed axial view of phase-contrast micro-CT slices depicting cervical-level human spinal column anatomy within a Thiel-embalmed specimen. Spinal cord: SC, epidural space: eDS, spinal meninges: SM, dorsal and ventral nerves: dN, vN, dorsal root ganglia: dRG, vertebral body: VB. Red asterisks label vasculature perfused with ANGIOFIL (iodinated contrast agent).
The volumetric nature of the collected CT data permitted 2D to 3D visualisations and explorations of micrometric spinal anatomy without the need for sample sectioning. Imaging of the Thiel-embalmed samples demonstrated macroscopic spinal column anatomy and inter-tissue boundaries, and resulted in soft-tissue contrast within the vertebral canal, and in the rendering of gross spinal cord anatomy, spinal meninges, spinal spaces, and contrast agent-enhanced spinal vasculature alongside surrounding vertebral bone structure (Figure 1). It also demonstrated the feasibility of concurrent high-contrast soft-tissue and bone-tissue morphologic visualisations with the same X-ray phase-contrast micro-CT data, with image gray levels optimisable alternatively for soft tissue or for bone structure visualisations. The extracted and formalin-fixed human cadaveric spinal cord samples lead to intra-cord grey versus white matter contrast and, notably, to the contrast agent-free visualisation of micrometre-scale intramedullary vascular and cellular structures. These data permitted detailed examination of the medullary blood supply network, including both its central and peripheral systems, and of neuron cell somas populating anterior grey column nuclei (Figure 2).
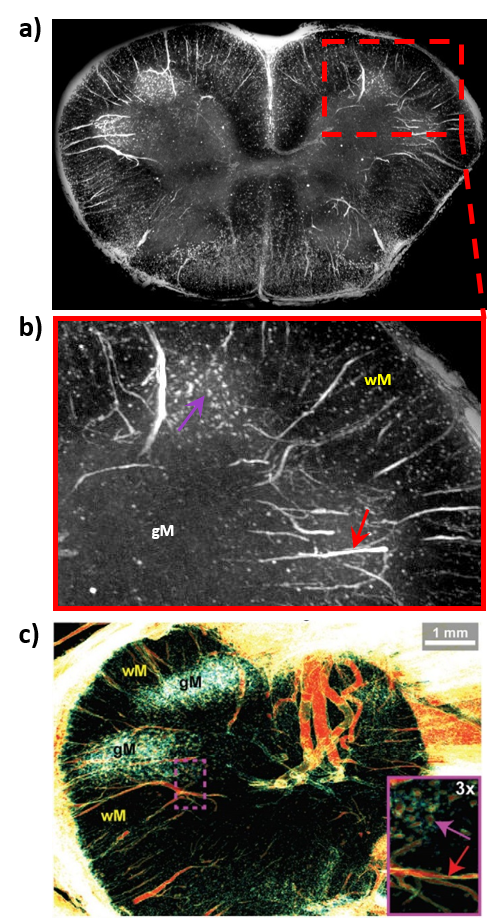
Fig. 2: a) Two-dimensional maximum intensity projection and (b) zoomed view, of an extracted formalin-fixed human spinal cord specimen, obtained from 100 consecutive slices of the same phase-contrast micro-CT 3D image stack. c) 3D rendering (and zoom, inset) of intramedullary vascular and cellular microanatomy. Grey matter: gM, white matter: wM, vascular features (red arrows), cellular features (magenta arrows).
This proof-of-concept study demonstrates that postmortem X-ray phase-contrast micro-CT provides unique knowledge of vertebral bone, soft tissue, and vasculature in 3D at high spatial resolution within a single CT image. This makes it attractive for multiscale tissue-preserving anatomic imaging work in human spinal cord and column specimens and provides an alternative or complementary method to traditional histologic or virtual 3D imaging techniques.
Principal publication and authors
High-spatial-resolution three-dimensional imaging of human spinal cord and column anatomy with postmortem X-ray phase-contrast micro-CT, G.E. Barbone (a,b), A. Bravin (c), A. Mittone (c), S. Grosu (b), J. Ricke (b), G. Cavaletti (d,e), V. Djonov (f), P. Coan (a,b), Radiology (2020); https://doi.org/10.1148/radiol.2020201622.
(a) Faculty of Physics, Ludwig-Maximilians-Universität München (Germany)
(b) Department of Radiology, University Hospital, Ludwig-Maximilians-Universität München (Germany
(c) ESRF
(d) School of Medicine and Surgery, University of Milano-Bicocca (Italy)
(e) Milan Center for Neuroscience, University of Milano-Bicocca (Italy)
(f) Institute of Anatomy, University of Bern (Switzerland)
References
[1] J.D. Van Horn & A.W. Toga, Brain Imaging Behav. 8(2), 323-331 (2014).
[2] O.J. Arthurs et al., Clin. Radiol. 70(8), 872-880 (2015).
[3] M. Kampschulte et al., RöFo 188(2), 146-154 (2016).
[4] F. Arfelli et al., Radiology 215(1), 286-293 (2000).
[5] H. Hetterich et al., Radiology 271(3), 870-878 (2014).
[6] A. Horng et al., Invest. Radiol. 49(9), 627-634 (2014).
[7] A. Khimchenko et al., Adv. Sci. (Weinh) 5(6), 1700694 (2018).
[8] G.E. Barbone et al., J. Neurosci. Methods 339, 108744 (2020).
[9] M. Fratini et al., Sci. Rep. 5(1), 8514 (2015).



